Artificial sphincter insertion
Definition
Artificial sphincter insertion surgery is the implantation of an artificial valve in the genitourinary tract or in the anal canal to restore continence and psychological well being to individuals with urinary or anal sphincter insufficiency that leads to severe urinary or fecal incontinence.
Purpose
This procedure is useful for adults and children who have severe incontinence due to lack of muscle contraction by either the urethral sphincter or the bowel sphincter. The primary work of the lower urinary tract and the colon is the storage of urine and waste, respectively, until such time as the expulsion of urine or feces is appropriate. These holding and expelling functions in each system require a delicate balance of tension and relaxation of muscles, especially those related to conscious control of the act of urination or defecation through the valve-like sphincter in each system. Both types of incontinence have mechanical causes related to reservoir adequacy and sphincter, or "gatekeeper" control, as well as mixed etiologies in the chemistry, neurology, and psychology of human makeup. The simplest bases of incontinence lie in the mechanical components of reservoir mobility and sphincter muscle tone. These two factors receive the most surgical attention for both urinary and fecal incontinence.
Urinary sphincter surgery
There are four sources of urinary incontinence related primarily to issues of tone in pelvic, urethral, and sphincter muscles. Most urinary incontinence is caused by leakage when stress is applied to the abdominal muscles by coughing, sneezing, or exercising. Stress incontinence results from reduced sphincter adequacy in the ability to keep the bladder closed during movement. Stress incontinence can also be related to the mobility of the urethra and whether this reservoir for urine tilts, causing spilling of urine. The urethral cause of stress incontinence is treated with other surgical procedures. A second form of incontinence is urge incontinence. It relates to sphincter overactivity, or sphincter hyperflexia, in which the sphincter contracts uncontrollably, causing the patient to urinate, often many times a day. Finally, there is urinary incontinence due to an inadequately small urethra that causes urine overflow. This is known as overflow incontinence and can often be treated with augmentation to the urethra to increase its size.
Only severe stress incontinence related to sphincter adequacy can benefit from the artificial urinary sphincter.
Implantation surgery related to urinary sphincter incompetence is also called artificial sphincter insertion or inflatable sphincter insertion. The artificial urinary sphincter (AUS) is a small device placed under the skin that keeps pressure on the urethra until there is a decision to urinate, at which point a pump allows the urethra to open and urination commences. Since the 1990s, advances in prostate cancer diagnosis and surgery have resulted in radical prostatectomies being performed, with urinary incontinence rates ranging from 3–60%. The AUS has become a reliable treatment for this main source of urinary incontinence in men. Women with intrinsic sphincter deficiency, or weakened muscles of the sphincter, also benefit from the AUS. However, the use of AUS with women has declined with advances in the use of the sub-urethral sling due to its useful "hammock" effect on the sphincter and its high rates of continence success. Women with neurologenic incontinence can benefit from the AUS.
Artificial anal sphincter surgery
Fecal incontinence is the inability to control bowel function. The condition can be the result of a difficult childbirth, colorectal disease such as Crohn's disease, accidents involving neurological injuries, surgical resection for localized cancer, or by other neurological disorders. Severe fecal incontinence may, depending upon the underlying disease, require surgical intervention that can include repair of the anal sphincter, colostomy , or replacement of the anal sphincter. Artificial anal sphincter is a very easy-to-use device implanted under the skin that mimics the function of the anal sphincter.
Demographics
Artificial urinary sphincter surgery
According to the Agency for Health Care Policy and Research, urinary incontinence affects approximately 13 million adults. Men have incontinence rates that are much lower than women, with a range of 1.5–5%, compared to women over 65 with rates of almost 50%. In older men, prostate problems and their treatments are the most common sources of incontinence. Incontinence is a complication in nearly all male patients for the first three to six months after radical prostatectomy. A year after the procedure, most men regain continence. Stress incontinence occurs in 1–5% of men after the standard treatment for severe benign prostatic hyperplasia.
Artificial anal sphincter surgery
According to the National Institute of Diabetes & Digestive & Kidney Diseases (NIDDK), more than 6.5 million Americans have fecal incontinence. Fecal incontinence affects people of all ages. It is estimated that over 2% of the population is affected by fecal incontinence. Many cases are never reported. Community-based studies reveal that 30% of patients are over the age of 65, and 63% are female. According to one study published in the American Journal of Gasteronology, only 34% of incontinent patients have ever mentioned their problem to a physician, even though 23% wear absorbent pads, 12% are on medications, and 11% lead lives restricted by their incontinence. Women are more than five times as likely as men to have fecal incontinence, primarily due to obstetric injury, especially with forceps delivery and anal sphincter laceration. Fecal incontinence is frequent in men who have total and subtotal prostatectomies. Fecal incontinence is not a part of aging, even though it affects people over 65 in higher numbers than other populations.
Description
Artificial urinary sphincter surgery
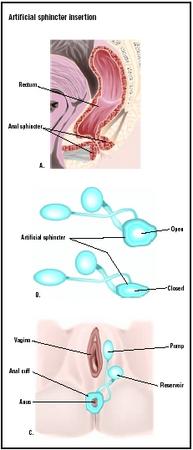
The artificial urinary sphincter is an implantable device that has three components:
- an inflatable cuff
- a fluid reservoir (balloon)
- a semiautomatic pump that connects the cuff and balloon
Open surgery is the major form of surgery for the implant. Infections are minimized by sterilization of the urine preoperatively and preoperative bowel preparation. The pelvic space is entered from the abdomen or from the vagina, with general anesthesia for the patient. Broad-spectrum antibiotics are given intravenously and at the site of small incisions for the device. A urinary catheter is put into place. The cuff is implanted around the bladder neck and secured and passed through the rectus muscle and anterior fascia to be connected later to the pump. A space is fashioned to hold the balloon in the pubic region, and a pump is placed in a pouch below the abdomen. The artificial urinary sphincter is activated only after six to eight weeks to allow healing from the surgery. The patient is trained in the use of the device by understanding that the cuff remains inflated in its "resting state," and keeps the urethra closed by pressure, allowing continence. Upon the decision to urinate, the patient temporarily deflates the cuff by pressing the pump. The urethra opens and the bladder empties. The cuff closes automatically.
Artificial anal sphincter surgery
The artificial anal sphincter is an implantable device that has three components:
- an inflatable cuff
- a fluid reservoir (balloon)
- a semiautomatic pump that connects the cuff and balloon
In open abdominal surgery, the implant device is placed beneath the skin through small incisions within the pelvic space. One incision is placed between the anus and the vagina or scrotum, and the inflatable cuff is put around the neck of the anal sphincter. A second incision at the lower end of the abdomen is used to make a space behind the pubic bone for placement of the balloon. The pump is placed in a small pocket beneath the labia or scrotum, using two incisions. The artificial anal sphincter is activated only after six to eight weeks to allow healing from the surgery. The patient is trained in the use of the device by understanding that the anal cuff remains inflated in its "resting state," and keeps the anal canal closed by pressure, allowing continence. Upon the decision to have a bowel movement, the patient temporarily deflates the cuff by pressing the pump and fecal matter is released. The balloon re-inflates after the movement.
Diagnosis/Preparation
Artificial urinary sphincter surgery
Patients must be chosen carefully, exhibit isolated sphincter deficiency, and be motivated and able to work with the device and its exigencies. To characterize the condition to be treated and to determine outcomes, full clinical, urodynamic, and radiographic evaluations are necessary. The ability to distinguish mobility of the urethra as the cause of incontinence from sphincter insufficiency is difficult, but very important in the decision for surgery. A combination of pelvic examination for urethral hypermobility and a leak-point pressure as measured by coughing or other abdominal straining has been shown to be very effective in identifying the patient who needs the surgical implant. Visual examination of the bladder with a cystoscope is very important in the preoperative evaluation for placement of the sphincter. Urethral and bladder conditions found by the examination should be addressed before implantation. Previous reconstruction or repair of the urethra may prevent implantation of the cuff. In open abdominal surgery, the implant surgery uses preventive infection measures that are very important, including sterilization of the urine preoperatively with antibiotics, the cleansing of the intestines from fecal matter and secretions through laxatives immediately prior to surgery, and antibiotic treatment and vigorous irrigation of the wound sites.
Artificial anal sphincter surgery
Since only a limited number of patients with fecal incontinence would benefit from an artificial sphincter, it is very important that a thorough examination be performed to distinguish the causes of the incontinence. A medical history and physical, as well as documented entries or an incontinence diary are crucial to the diagnosis of fecal incontinence. The physical exam usually includes a visual inspection of the anus and the area lying between the anus and genitals for hemorrhoids, infections, and other conditions. The strength of the sphincter is tested by the doctor probing with a finger to test muscle strength.
Medical tests usually include:
- Anorectal manometry. This is a long tube with a balloon on the end that is inserted in the anus and rectum to measure the tightness of the anal sphincter and the ability to respond to nerve firings.
- Anorectal ultrasonography. This test also includes an insertion of a small instrument into the anus with a video screen that produces sound waves, picturing the rectum and anus.
- X rays. A substance called barium is used to make the rectum walls visible to x ray. This liquid is swallowed by the patient before the test.
- Anal electromyography. This test uses the insertion of tiny needle electrodes into muscles around the anus and tests for nerve damage.
Aftercare
Artificial urinary sphincter surgery
Surgery requires a few days of hospitalization. Oral and intravenous pain medications are administered, along with postoperative antibiotics. A general diet is available, usually on the evening of surgery. When the patient is able to walk, the urethral catheter is removed. Patients are discharged on the second day postoperatively, unless they have had other procedures and need extra recovery time. Patients may not lift heavy objects or engage in strenuous activity for approximately six weeks. After six to eight weeks, the patient returns to the physician for training in the use of the implant device.
Artificial anal sphincter surgery
Surgery hospitalization requires a few days with dietary restrictions and anti-diarrheal medicine to bind the bowels. Antibiotics are administered to lower the risk of infection, and skin incisions are cleaned frequently. Patients may not lift heavy objects or engage in strenuous activity for approximately six weeks. After the body has had time to heal over six to eight weeks, the patient returns to the physician for training in the use of the pump. Two or three sessions are required and after the training, the patient is encouraged to lead as normal a life as possible.
Normal results
Artificial urinary sphincter surgery
One problem with the urinary sphincter implant is failure. If the device fails, or the cuff erodes, the surgery must be repeated. In a study published in 2001, 37% of women had the implant after an average of seven years, but 70% had the original or a replacement and 82% were continent. Studies on men report similar findings. Malfunction has improved with advances in using a narrower cuff. In one large study encompassing one surgeon over 11 years, the re-operative rate of AUS related to malfunction in men was 21%. Over 90% of patients were alive with a properly functioning device.
Another problem with the surgery is urinary voiding. This may be difficult initially due to postoperative edema caused by bruising of the tissue. In the majority of cases, urination occurs after swelling has receded.
AUS is a good alternative for children. The results of AUS in children range from 62–90%, with similar rates for both girls and boys.
Artificial anal sphincter surgery
Anal sphincter implant surgery has been successfully performed for many years. The device most often used has a cumulative failure rate of 5% over 2.5 years. The long-term functional outcome of artificial anal sphincter implantation for severe fecal incontinence has not been determined. However, adequate sphincter function is recovered in most cases, and the removal rate of the device is low. Most of the good results are dependent upon careful patient selection and appropriate surgical and operative management with a highly experienced surgical team .
Morbidity and mortality rates
Artificial urinary sphincter surgery
Infection has been a frequent and serious complication of surgery, not only because of the infection per se, but also because infection can cause erosion of the urethra or bladder neck under the implant. The infection may actually worsen the incontinence. The overall infection rate with AUS implants is 1–3%. Because of interactions between the host and the foreign body represented by the implant, infections can occur soon after the surgery, or months and even years later. New techniques using antibiotics and skin preparations have improved infection rates considerably.
Artificial anal sphincter surgery
This surgery is for a limited number of patients who have isolated sphincter deficiency. Patients must be chosen who have little co-morbidity (serious illnesses) and can be trained in the use of the pump. Although it is a fairly simple operation, some researchers report a 30% infection rate.
Alternatives
Artificial urinary sphincter surgery
Milder forms of urinary incompetence can be treated with changes in diet, evaluation of medications, and the use of antidepressants and estrogen replacement, as well as bladder training and pelvic muscle strengthening. However, sphincter deficiency, unlike incontinence caused by urethral mobility, requires a substitute for the sphincter contraction by implant or by auxiliary tissue. If AUS cannot treat sphincter deficiency, the sling or "hammock" procedure is a good second choice. It brings tightness to the sphincter by using tissue under the urethra to increase contractual function. The sling procedure is already preferred over the AUS for women.
Artificial anal sphincter surgery
Milder forms of fecal incontinence are being treated by changes in diet and the use of certain bowel-binding medications. For some forms of mild fecal incontinence, special forms of exercise can help to strengthen and tone the pelvic floor muscles, along with providing biofeedback to train the muscles to work with an appropriate schedule. Only after these measures have been tried, including the use of pads, is the patient counseled on the benefits of an anal sphincter implant.
Resources
books
Walsh, P., et al. Campbell's Urology, 8th Edition. St. Louis: Elsevier Science, 2000.
periodicals
Michot, F. "Artificial Anal Sphincter in Severe Fecal Incontinence: Outcome of Prospective Experience with 37 Patients in One Institution." Annals of Surgery, Vol. 237, No. 1 (January 1, 2003): 52–56.
Rotholtz, N. A., and S. D. Wexner. "Surgical Treatment of Constipation and Fecal Incontinence." Gastroenterology Clinics, Vol. 30, No. 01 (March 2001).
organizations
American Society of Colon and Rectal Surgeons. 85 W. Algonquin Rd., Suite 550, Arlington Heights, IL 60005. http://www.fascrs.org. .
National Institute of Diabetes and Digestive and Kidney Diseases. (800) 891-5390 (kidney); (800) 860-8747 (diabetes); (800) 891-5389 (digestive diseases). http://www.niddk.nih.gov. .
National Association of Incontinence. http://www.nafc.org. .
other
Fecal Incontinence. National Institute of Diabetes & Digestive & Kidney Diseases (NIDDK). http://www.niddk.nih.gov/health/digest/pubs/fecalincon/fecalincon.htm .
Incontinence in Men. Health and Age. http://www.healthand age.com. .
Urinary Incontinence. WebMD Patient Handout. http://www.MD-consult.com. .
Urinary Incontinence in Women. National Institute of Diabetes & Digestive & Kidney Diseases (NIDDK). http://www.niddk.nih.gov/health/urolog/pubs/uiwomen/uiwomen.htm. .
Nancy McKenzie, PhD
WHO PERFORMS THE PROCEDURE AND WHERE IS IT PERFORMED?
Implantation surgery is performed in a hospital operating room by urologic surgeons specially trained for implantation of the artificial sphincter in the urinary or anal tracts. Successful surgery depends upon very experienced surgeons.
QUESTIONS TO ASK THE DOCTOR
- How many implantation surgeries have you performed?
- What is your rate of device removal in the patients you have treated?
- How likely is infection after surgery?
- How likely is infection to occur long term?
What new possible medical help is forthcoming for ED?
Sid Fetter
I am hopeful that a lower end GI transplant will put my life back in order, or some other type of device.. I have the means necessary to pay out of pocket if my issuance feels I don't need the procedure. Doc Presents office notified me that there are many studies being conducted. I have no other bowel issues. It was contained only in the very end of my large bowel. Top end of the rectum , sigmoid, and anus. I have all my intestine and it is in very good health. I have been part of the CCFA for years now. I am friends with Doc Sacker, Doc Bayles at John Hopkins, and part of Doc Presents office since 1993. Doc Janowiz I befriended before his death.
I am honestly in need of help and do not wont to spend the rest of my life using a Colostomy bag. I feel a transplantation may be the answer. Please inform me if you have any information on this matter. Other programs being offered. There must be some type of study going on some place.
Please help me,
Gary Mazza 78 weiner st staten island ny 10309 or Art my father please call 718 356 6460 917 886 1108
where we can do this operation for him specially in Europe if there a such clinic con do ?
thanks
Is this urgency due to bruising of the tissue? Will it quieten down after about 3-6 months?
He has a clinic appointment at the hospital UVLH end of April.
Thank you
I live in southern Brazil
I ostomy pouch 20 years, and can not stand anymore.
Never, never I had job and girlfriend and social life because of the stoma.
It is the realization of my life doing the implant surgery anal sphincter
I used the google translator to write this message because I can not speak English.
love to have contact with the drug class that has experience in this type of surgery: anal sphincter implantation.
thank you
10 October 2012 the 19 55 'time in Brazil'
Please let me know where is this hospital. Appreciate your cooperate. Await for your response please.