Pacemakers
Definition
A pacemaker is a surgically implanted electronic device that regulates a cardiac arrhythmia.
Pacemakers are most frequently prescribed when the heartbeat decreases under 60 beats per minute at rest (severe symptomatic bradycardia). They are also used in some cases to slow a fast heart rate over 120 beats per minute at rest (tachycardia).
Demographics
The population for pacemaker implant is not limited by age, sex, or race. Over 100,000 pacemakers are implanted per year in the United States. The occurrence is more frequent in the elderly with over 85% of implants received by those over age 65. A history of myocardial infarction (heart attack), congenital defect, or cardiac transplant also increases the likelihood of pacemaker implant.
Description
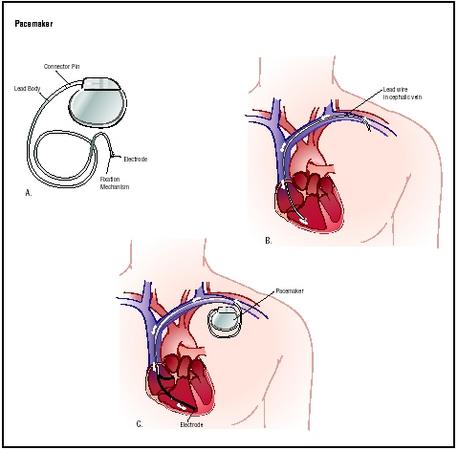
Approximately 500,000 Americans have an implantable permanent pacemaker device. A pacemaker implantation is performed under local anesthesia in a hospital by a surgeon assisted by a cardiologist. An insulated wire called a lead is inserted into an incision above the collarbone and guided through a large vein into the chambers of the heart. Depending on the configuration of the pacemaker and the clinical needs of the patient, as many as three leads may be used in a pacing system. Current pacemakers have a double, or bipolar, electrode attached to the end of each lead. The electrodes deliver an electrical charge to the heart to regulate heartbeat. They are positioned on the areas of the heart that require stimulation. The leads are then attached to the pacemaker device, which is implanted under the skin of the patient's chest.
Patients undergoing surgical pacemaker implantation usually stay in the hospital overnight. Once the procedure is complete, the patient's vital signs are monitored and a chest x ray is taken to ensure that the pacemaker and leads are properly positioned.
Modern pacemakers have sophisticated programming capabilities and are extremely compact. The smallest weigh less than 13 grams (under half an ounce) and are the size of two stacked silver dollars. The actual pacing device contains a pulse generator, circuitry programmed to monitor heart rate and deliver stimulation, and a lithium iodide battery. Battery life typically ranges from seven to 15 years, depending on the number of leads the pacemaker is configured with and how much energy the pacemaker uses. When a new battery is required, the unit can be exchanged in a simple outpatient procedure.
A temporary pacing system is sometimes recommended for patients who are experiencing irregular heartbeats as a result of a recent heart attack or other
Diagnosis/Preparation
Patients being considered for pacemaker implantation will undergo a full battery of cardiac tests, including an electrocardiogram (ECG) or an electrophysiological study or both, to fully evaluate the bradycardia or tachycardia.
The symptoms of fatigue and lightheadedness that are characteristic of bradycardia can also be caused by a number of other medical conditions, including anemia. Certain prescription medications can also slow the heart rate. A doctor should take a complete medical history and perform a full physical work-up to rule out all non-cardiac causes of bradycardia.
Patients are advised to abstain from eating six to eight hours before the surgical procedure. The patient is usually given a sedative to help him or her relax for the procedure. An intravenous (IV) line will also be inserted into a vein in the patient's arm before the procedure begins in case medication or blood products are required during the insertion.
Aftercare
After an implant without complications the patient can expect a hospital stay of one to five post-procedure days. Pacemaker patients should schedule a follow-up visit with their cardiologist approximately six weeks after the surgery. During this visit, the doctor will make any necessary adjustments to the settings of the pacemaker. Pacemakers are programmed externally with a handheld electromagnetic device. Pacemaker batteries must be checked regularly. Some pacing systems allow patients to monitor battery life through a special telephone monitoring service that can read pacemaker signals.
Patients with cardiac pacemakers should not undergo a magnetic resonance imaging (MRI) procedure. Devices that emit electromagnetic waves (including magnets) may alter pacemaker programming or functioning. A 1997 study found that cellular phones often interfere with pacemaker programming and cause irregular heart rhythm. However, advances in pacemaker design and materials have greatly reduced the risk of pacemaker interference from electromagnetic fields.
Risks
Because pacemaker implantation is an invasive surgical procedure, internal bleeding, infection, hemorrhage, and embolism are all possible complications. Infection is more common in patients with temporary pacing systems. Antibiotic therapy given as a precautionary measure can reduce the risk of pacemaker infection. If infection does occur, the entire pacing system may have to be removed.
The placing of the leads and electrodes during the implantation procedure also presents certain risks for the patient. The lead or electrode could perforate the heart or cause scarring or other damage. The electrodes can also cause involuntary stimulation of nearby skeletal muscles.
A complication known as pacemaker syndrome develops in approximately 7% of pacemaker patients with single-chamber pacing systems. The syndrome is characterized by the low blood pressure and dizziness that are symptomatic of bradycardia. It can usually be corrected by the implantation of a dual-chamber pacing system.
Normal results
Pacemakers that are properly implanted and programmed can correct a patient's arrhythmia and resolve related symptoms.
Morbidity and mortality rates
In the United States, patients experience complications in 3.3% and 3.8% of cases, with those over 65 years of age demonstrating a slightly higher complication rate of 6.1%. The most common complications include lead dislodgement, pneumothorax (collapsed lung), and cardiac perforation. The risk of death is less then 0.5% throughout the course of the hospital stay.
Resources
books
DeBakey, Michael E. and Antonio Gotto Jr. The New Living Heart. Holbrook, MA: Adams Media Corporation, 1997.
periodicals
Gregoratas, Gabriel, et al. "ACC/AHA Guidelines for Implantation of Pacemakers and Antiarrhythmia Devices." Journal of the American College of Cardiology 31 (April 1998): 1175–209.
Link, Mark S, et al. "Complications of Dual Chamber Pacemaker Implantation in the Elderly." Journal of Interventional Cardiac Electrophysiology 2 (1998): 175–179.
organizations
American Heart Association. 7320 Greenville Ave. Dallas, TX 75231. (214) 373-6300. http://www.americanheart.org .
Paula Anne Ford-Martin Allison J. Spiwak, MSBME
WHO PERFORMS THE PROCEDURE AND WHERE IS IT PERFORMED?
Pacemaker implants are performed by a cardiologist who has completed medical school and an additional internship and residency program. Additional training as an electrophysiologist may be acquired by the physician during the residency program. Specific training by the pacemaker manufacturer may also be acquired. Hospitals performing these procedures have access to cardiac catheterization facilities or operating rooms equipped with portable fluoroscopy units.
QUESTIONS TO ASK THE DOCTOR
- How many pacemaker implants has the physician performed?
- What type of pacemaker will be implanted, univentricular or biventricular, and how many of the specific procedure has the physician performed?
- How long will the expected hospital stay be?
- What precautions should be taken in the weeks following discharge from the hospital?
- What precautions will need to taken in day to day activities following pacemaker implant?
- When can normal daily, such as driving, exercise and work, activities be initiated?
- What will indicate that the pacemaker is failing and when should emergency care be sought?
- How long will the battery function and when should treatment to replace the device be sought?
- Is there special documentation I will need for air travel during security screenings?
- Will there be notification of manufacturer recalls?
by reading it we can fulll information about pacemaker
plz read its
2 min. after the surgery I was asking for coffee and a donut. Surgery perform at WELLSTAR HOSPITAL in Marietta, GA.
Thanks to the outstanding Doctors on the WELLSTAR Cardiovascular Center.
The doctors could not see the end of the peak of the electrode and wanted me to have slice scanner done but I was told that it was not possible because of irregular heart rhythm and they were afraid of damaging the electro leader by exposing it to the CT.
Mother continues to experience severe right arm pain. I know this is not a situation of "overuse" of the right arm. My mother was a college athlete and was beating me at tennis at age 60. She knows what a strained muscle feels like!
I am now losing confidence in the surgeon. What treatments are proven successful in treating this pain, which is related to pacemaker implant sugery?
Its been more than a month to the surgery and my father is feeling some pain in the left shoulder and left hand. The pacemaker is placed on the left side above the lungs.
After reading this post I can conclude that my father is suffering from one of the pacemaker complications i.e. embolism.
Is there a solution of it?
Many thanks x
Thanks for the reply it means alot, im having real yrouble finding any pictures of the procedure and dont no much about it at all. Really appreciate the advice thou i will make sure i get a supportive bra to take with me. Would it be possible for you to tell me what you no about the procedure please x
Many thanks from vicky x
The Manufacturer pulled the wire off the market, and told all Doctors to stop installing the Questionable wire that was causing injury and deaths.
During the insertion of the new up-graded Pace/Defibr device, the Doctor attached the original "Recalled" lead wire to the new upgraded device.
Should I be concerned?
In January 2012 it was determined that the battery, which should have lasted 5-7 years, would not last much longer as the voltage being used is inordinately high. Not sure what the cause is.
My 85 year old relative has Parkinson's and dementia, has had a stroke, subdural hematoma, lymphoma and had a mitral valve repair years ago that still has a slight regurge .
Having said this, I feel it is risky to have new leads put in; can the device and or just the battery be replaced and connected to the same leads, even if there is some scar tissue?
Please advise.
Severe LV Dysfunction. The level of efficiency is only 15% NO PAH.Type 2 diabetes & pre renal & diabetic CKD. Past history of H/O DVT with PTE on chronic anti coagulation.
Could you please advise whether going for an pace maker implant will help & improve my mothers heart condition.
Awaiting your favourable reply & please help on humanitarian grounds.
My dad had a infection blood poisoning. Take a stand the pacemaker killed my daddy. The pacemaker will beat for ever and my dadddy was dead. The doctors lie this is just for money they lied to my dad. When you think about life why wouldnt anybody want a pacemaker. More people die instead of getting healed. My dad is in a better place even without that stupid ass pacemaker wat a piece of crap. Very angry daughter...
Last Thursday i got a reveal recorder inserted into me and they warned me that i will be in discomfort for the next 24 hours but i am still in discomfort now, is that normal?
We have visited the doctor several times but the problem still exists...
My question is I got my new replacement this last week and was told the leads were going bad. Why wouldn't they replace them before problems start and what is the mortality rate of the different methods of lead wire replacements?
Nitu Phukan
Assam
I just removed the dressing last night and discovered that what they in fact did was cut an inch and a half below the original scar. No one said anything when I was in the OR and when they were putting the dressing on I was completely out of sedation. Since I was already out of sedation I was able to bypass recovery and went directly back to Day surgery.
I am very sensitive about the scars on my body and already have a lot from heart surgery and the original pacemaker being put in and another surgery that was unrelated.
Seeing as I am only 42 years old and will have many other replacements, does this mean I am going to end up with a new scar everytime I have it changed?
can anyone tell me is this a diabetic /defib problem or something else
even I take Insulin. Sometime I regret why I got this pace maker in my body. I think my body did not like the pacemaker. I don't know what to do.
Please advice. Thanks, Indra
I hope this helps, good luck
I had a light breakfast at 6am was taken to hospital for 8am. Chest shaved, blood pressure taken ,I was second on the list the chap before me was having a new pacemaker as the old one was 11 years old and time for a new one. I went for mine at 10:15 am was back at 11:30am for cup of coffe and sandwich (very nice) then i was taken for chest Xray.
The pacemaker was then programmed. I was told the do's and dont's and after the Xray results was allowed to go home at 3:15 pm. Must say I feel great.
At the same time felt slightly short of breath and still have sharp pain in center of my back.
My question is, has anyone else been through this, and did you choose to remove the old unit and leads? One dr said no, don't bother. Another said we should. the third said it was our choice. So, I'd rather not put her thru an additional surgery, but hate leaving that stuff in her body, too.
Has anyone experienced this or know anything about this vibration I have in my body. All my blood work, scans, ekgs, echos, xrays are normal. I feel good other than these vibrations.
,
so they removed it and put another on the right side of her chest
Now 5 days later, both of her arms have turned black color Her breast are also black
SHe can hardly use her arms for any thing thing like cooking or even pulling up underpants
thanks we need help and pryaers.
Can you shareyour experiences and what shall she do she is 7 and sort of frail