Tubal ligation
Definition
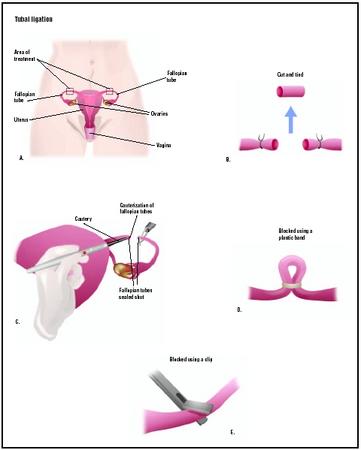
Tubal ligation is a permanent voluntary form of birth control (contraception) in which a woman's fallopian tubes are surgically cut or blocked off to prevent pregnancy.
Purpose
Tubal ligation is performed in women who want to prevent future pregnancies. It is frequently chosen by women who do not want more children, but who are still sexually active and potentially fertile, and want to be free of the limitations of other types of birth control. Women who should not become pregnant for health concerns or other reasons may also choose this birth control method.
Demographics
Tubal ligation is one of the leading methods of contraception, having been chosen by over 10 million women in the United States—about 15% of women of reproductive age. The typical tubal ligation patient is over age 30, is married, and has had two or three children.
Description
Tubal ligation, or getting one's "tubes tied," refers to female sterilization, the surgery that ends a woman's ability to conceive. The operation is performed on the patient's fallopian tubes. These tubes, which are about 4 in (10 cm) long and 0.2 in (0.5 cm) in diameter, are found on the upper outer sides of the uterus. They open into the uterus through small channels. It is within the fallopian tube that fertilization, the joining of the egg and the sperm, takes place. During tubal ligation, the tubes are cut or blocked in order to close off the sperm's access to the egg.
Normally, tubal ligation takes about 20–30 minutes, and is performed under general anesthesia, spinal anesthesia,
Tubal ligation should be postponed if the woman is unsure about her decision. While the procedure is sometimes reversible, it should be considered permanent and irreversible. As many as 10% of sterilized women regret having had the surgery, and about 1% seek treatment to restore their fertility.
The most common surgical approaches to tubal ligation include laparoscopy and mini-laparotomy. In a laparoscopic tubal ligation, a long, thin telescope-like surgical instrument called a laparoscope is inserted into the pelvis through a small cut about 0.5 inches (1 cm) long near the navel. Carbon dioxide gas is pumped in to help move the abdominal wall to give the surgeon easier access to the tubes. Often the surgical instruments are inserted through a second incision near the pubic hair line. An instrument may be placed through the vagina to hold the uterus in place.
In a mini-laparotomy, a 1.2–1.6 in (3–4 cm) incision is made just above the pubic bone or under the navel. A larger incision, or laparotomy, is rarely used today. Tubal ligation can also be performed at the time of a cesarean section .
The tubal ligation itself is performed in several ways:
- Electrocoagulation. A heated needle connected to an electrical device is used to cauterize or burn the tubes. Electrocoagulation is the most common method of tubal ligation.
- Falope ring. In this technique, an applicator is inserted through an incision above the bladder and a plastic ring is placed around a loop of the tube.
- Hulka clip. The surgeon places a plastic clip across a tube held in place by a steel spring.
- Silicone rubber bands. A band placed over a tube forms a mechanical block to sperm.
Tubal ligation costs about $2,000 when performed by a private physician, but is less expensive when performed at a family planning clinic. Most insurance plans cover treatment costs.
Diagnosis/Preparation
Preparation for tubal ligation includes patient education and counseling. Before surgery, it is important that the woman understand the permanent nature of tubal ligation as well as the risks of anesthesia and surgery. Her medical history is reviewed, and a physical examination and laboratory testing are performed. The patient is not allowed to eat or drink for several hours before surgery.
Aftercare
After surgery, the patient is monitored for several hours before she is allowed to go home. She is instructed on care of the surgical wound, and what signs to watch for, such as fever, nausea, vomiting, faintness, or pain. These signs could indicate that complications have occurred.
Risks
While major complications are uncommon after tubal ligation, there are risks with any surgical procedure. Possible side effects include infection and bleeding. After laparoscopy, the patient may experience pain in the shoulder area from the carbon dioxide used during surgery, but the technique is associated with less pain than mini-laparotomy, as well as a faster recovery period. Mini-laparotomy results in a higher incidence of pain, bleeding, bladder injury, and infection compared with laparoscopy. Patients normally feel better after three to four days of rest, and are able to resume sexual activity at that time.
The possibility for treatment failure is very low—fewer than one in 200 women (0.4%) will become pregnant during the first year after sterilization. Failure can happen if the cut ends of the tubes grow back together; if the tube was not completely cut or blocked off; if a plastic clip or rubber band has loosened or come off; or if the woman was already pregnant at the time of surgery.
Normal results
After having her tubes tied, a woman does not need to use any form of birth control to avoid pregnancy. Tubal ligation is almost 100% effective for the prevention of conception.
Morbidity and mortality rates
About 1–4% of patients experience complications following tubal ligation. There is a low risk (less than 1%, or seven per 1,000 procedures) of a later ectopic pregnancy. Ectopic pregnancy is a condition in which the fertilized egg implants in a place other than the uterus, usually in one of the fallopian tubes. Ectopic pregnancies are more likely to happen in younger women, and in women whose tubes were closed off by electrocoagulation.
Rarely, death may occur as a complication of general anesthesia if a major blood vessel is cut. The mortality rate of tubal ligation is about four in 100,000 sterilizations.
Alternatives
There are numerous options available to women who wish to prevent pregnancy. Oral contraceptives are the second most common form of contraception—the first being female sterilization—and have a success rate of 95–99.5%. Other methods of preventing pregnancy include vasectomy (99.9% effective) for the male partner; the male condom (86–97% effective); the diaphragm or cervical cap (80–94% effective); the female condom (80–95% effective); and abstinence.
See also Vasectomy .
Resources
books
"Family Planning: Sterilization." Section 18, Chapter 246 in The Merck Manual of Diagnosis and Therapy , edited by Mark H. Beers, MD, and Robert Berkow, MD. Whitehouse Station, NJ: Merck Research Laboratories, 1999.
periodicals
Baill, I. C., V. E. Cullins, and S. Pati. "Counseling Issues in Tubal Sterilization." American Family Physician 67 (March 15, 2003): 1287-1294.
Kariminia, A., D. M. Saunders, and M. Chamberlain. "Risk Factors for Strong Regret and Subsequent IVF Request After Having Tubal Ligation." Australian and New Zealand Journal of Obstetrics and Gynaecology 42 (November 2002): 526-529.
organizations
American College of Obstetricians and Gynecologists. 409 12th St., SW, P. O. Box 96920, Washington, DC 20090-6920. http://www.acog.org .
Planned Parenthood Federation of America, Inc. 810 Seventh Ave., New York, NY, 10019. (800) 669-0156. http://www.plannedparenthood.org
other
Centers for Disease Control and Prevention. Fact Sheet: Risk of Ectopic Pregnancy after Tubal Sterilization , August 6, 2002 [cited March 1, 2003]. http://www.cdc.gov/nccdphp/drh/mh_ectopic.htm .
Planned Parenthood Federation of America. All About Tubal Sterilization . [cited March 1, 2003]. http://www.plannedparenthood.org/bc/allabouttubal.htm .
Planned Parenthood Federation of America. Facts About Birth Control , January 2001 [cited March 1, 2003]. http://www.plannedparenthood.org/bc/bcfacts1.html .
Mercedes McLaughlin Stephanie Dionne Sherk
WHO PERFORMS THE PROCEDURE AND WHERE IS IT PERFORMED?
Tubal ligation is generally performed by an obstetrician/gynecologist, a medical doctor who has completed specialized training in the areas of women's general health, pregnancy, labor and childbirth, prenatal testing, and genetics. The procedure is performed in a hospital or family planning clinic, and usually as an outpatient procedure.
QUESTIONS TO ASK THE DOCTOR
- How many tubal ligations do you perform each year?
- What method of ligation will you use?
- What form of anesthesia will be used?
- How long will the procedure take?
- What side effects or complications might I expect?
- What is your failure rate?
Admitedly, I was under a lot of mental stress the week before & during my cycle period. But I'm now ok yet no menses. Scared & hesitant to do a pregnancy test.
thank you!!!
being pregnant? my menstrual cycle never been this late its been 3weeks im scare to take a pregnancy test.. can i be pregnant?
I have had a period every month after that and now 9 yrs later I have not had a period in 89 days my brest have gotten huge and are tender I have gained a little weight and I am always tired I feel like I am pregnant but I really dont want to know if I am or not I was wondering what u thought.. Thank u
thanks
bridget davis
I was 30 yrs old with three children when my husband and I decided to get tubes tied. When I was 38 we reversed the tubal ligation and now have two more beautiful children. I am now 42 and who knows, maybe child number six will be on the way...children are a blessing of the Lord! If you think you want to reverse there are many website that will direct you how to go about it. I think a big one is called blessed arrows or somthing like that. Okay, take care all!
I do want a way to permenantly fix the risk of pregancy, but now I am scared that it may do more harm than good.
Thanks
Mary
THANK YOU..
pregnantagain.com ~ try this site
thank you!!!
Any Advice?
i
I have had more then enough bacterial infections, and Ive been with the same person the entire time (sexually). The doctors ingore my infections when I ask if its related to my surgery.
Also I have severe pain on my right side the morning of my period, the pain is so bad I fell like my ovary is going to burst. So I take Midol or something for pain, the doctors will not give me anything for the pain~~
I feel though that the clamps are the reason for my repeating bacterial infections.
I may have to have the clamps removed in the future???
I am a 48 year old woman who has had my tubes tied 17 years ago. Both of my breasts are extremely swollen and my left one has started with a clear liquid about a week ago and now the discharge is white and a bit clear. I haven't had tender breasts in years prior to my period. What is going on can I be pregnant after all these years.
thank you
Regards
Ramanpreet Singh
IN SEPT. 2010 I HAD THE GASTRIC BYPASS DONE
CAN THE BAND COME UNDONE AFTER HAVING THE GASTRIC BYPASS SURGERY AND IF SO WHEN AND HOW
MY HUSBAND AND I WANT TO HAVE ANOTHER BABY AND I HAVE TALKED TO A DOCTOR CLOSE BY ME AND THEY CAN DO IT JUST COST ME WHICH I DONT THINK IS THAT BAD OF A PRICE AND I HAVE APOINTMENT IN FEBRUARY TO SEE THE DOCTOR BUT I AM JUST WONDERING IF I NEED THE TUBAL REVERVSAL DONE OR IF THEY ARE ALREADY UNDONE CAUSE OF HAVING THE GASTRIC BYPASS DONE
IT JANUARY 2011 AND I JUST STARTED WAKING UP WITH HEADACHES AND FOR ABOUT 2 MONTHS I GET THIS FEELING NAUOS FOR A WEEK AND I HAVE BEEN TIRED ALOT
DID A HOME PREGNATE TEST CAME OUT NEGATIVE
PLEASE E-MAIL ME BACK ABOUT THIS: madpeck@yahoo.com (SOMERSET/CAMBRIA COUNTY)
AND I DO KNOW SOMEONE (DOCTOR) WHO DOES TUBAL REVERSAL AROUND HERE
What are the chances i could get pregnet without reversing it
also was wondering about artificial insemination using my eggs along with my husband sperm? is that a possibility?
There are plenty of other options for women who want kids in the future. Only get something permanent if you are absolutely sure that you are done having kids or never want to have kids. Also, talking to your OB/GYN is better than asking random people on the internet. Just saying.
The odds of you being pregnant are very slim and there are other possibilities as to why you're spotting and cramping. If it worries you that much, you can go buy a pregnancy test from a drugstore. If you really think you need to, visit an OB/GYN. They will be able to figure out what's going on much more accurately than I can.
Is There Any Chance Of Blood Clots ?
Admitedly, I was under a lot of mental stress the week before & during my cycle period. But I'm now ok yet no menses. Scared & hesitant to do a pregnancy test.
i had laprascopy done 2yrs back now i did not get my periods last month. i got the urine tested twice it gave negative result what should i do iam so confused we get things donbe to be away from worries now i'm more worriedf. kindly answer me
I had Tubal ligation during a c-section, after 4 weeks I felt aches, pain and tender mucsal all over my body, it lasted 3 days and it went away. The next day my baby had runny nose and coughs and became ill and had to be hospitalised because he wasn't breathing well. Could my illness be related to my baby's health because I am breast feeding?
My baby is 7 weeks now and I have the same aches and pain, I went to visit my family doctor and was informed I have infection from the operation. How sucessful is the Tubal ligation if there is an infection and what do I need to do to recover?
2. How would one know that she has ectopic pregnancy & which are the signs?
Thank you.
Sorry if the spelling is incorrect.
The only part of me that hurts from the surgery are the right side of my ribs from where they pumped air into my to work, it burned for awhile when I would go to the bathroom from where they put a device in me to hold the uterus still while they worked (all perfetly normal).
If everything goes great during the procedure, the dr knows what they are doing and you don't have any complications, you should only miss about a week of normal activity. 1 day for presurgery labs and paperwork, 1 day for surgery, 1 or 2 day of recovery and 1 day for follow-up apptointment.
Hope this information helps anyone that has questions about the procedure.
Def. Not wanting any more kids my husband is working all the time we barely see him and when we do he barely helps and financially wevhavent been doing so good i just feel that this wuld be the best decision and i just feel like i dont have patience for more kids aswell.. ive been giving it a lot of thought for the past 4months and im 100% sure that its what i want i just need to know if its possible for me to have my tubes cut and burnt at tye age of 23
and she get pregnant by penetration again
Anyway, for those with heavy bleeding, ask about endometrial ablation. It helps heavy bleeding. This is why I can't have babies safely. And a couple of people mentioned a brownish discharge, could be a symptom of endometriosis. Last, my dr said because if my endo, I might have to go back on the pill to regulate my cycles. Just thought I'd share. If you have questions about ablation or endo, feel free to e-mail me: kamikazekiki0212@yahoo.com.
I've had 4 laparoscopic surgeries for endometriosis. It usually takes me a week to recover. I think a tubal ligation is 2-3 days to recover (your insides aren't re-arranged). My last female surgery was ablation & I was down a week from healing.
Best Wishes!
thank you
Thanks :)
Thanks :)
I have had a laposcopic sterilization done on the 13th of June 2012
However i seem to be bleeding alot and it last up to 7 days
Last month i got a period that lasted 7 days and then i skipped a week and now we are in the new month and again i have my periods ,i know its a new month but it is only a week apart
Is this normal ?
How long will i be bleeding so heavy ?
Do i need to worry ?
Do i have to take another contriceptive to regulate my periods or should i just let nature takes it course
Pls help ..i am a bit worried though
I am very happy that I had the surgery, but it seems there are a lot of women that regret it. My surgeon said it is reversible but not always does it work. So as I said, be 100% sure before:)
do I still get pregnant?
I had my tubes tired in July after giving birth but did not see my period for two months. Is it normal or do I have to go for a pregnancy test again?