Cochlear implants
Definition
A cochlear implant is a small, complex electronic device used to treat severe to profound hearing loss. It is surgically implanted underneath the skin behind the patient's ear.
Purpose
A cochlear implant delivers useful auditory signals from the environment to the patient by electronically bypassing nonfunctional parts of the ear and directly stimulating the auditory nerve. Unlike a hearing aid, it does not merely amplify sound. Instead, an implant increases the amount of nervous response to sound. Although it does not restore normal hearing, the additional input provided by the implant often improves sound detection and increases speech understanding.
Description
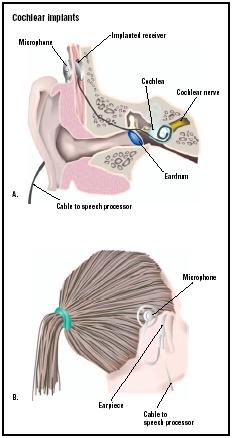
Normal hearing occurs because sound travels from the outer ear into the ear canal and vibrates the eardrum. The vibration is carried through the middle ear by three small bones attached to the eardrum and on to a fluid-filled part of the inner ear called the cochlea. Movement in the cochlear fluid is transferred to hair fibers within the cochlea. The movement of these hair cells stimulates nerve cells called ganglion cells that send an electrical current to the auditory nerve. In turn, the nerve carries the current to the brain, where the electrical stimulation is recognized as sound.
A common cause of hearing loss is damage to the hair cells within the cochlea. This kind of deafness, called sensorineural deafness, can often be treated with cochlear implants. This is particularly true if damage to the hair cells is not accompanied by damage to the auditory nerve itself. As of 2002, it is estimated that over 35,000 individuals have received cochlear implants.
Cochlear implants consist of internal and external parts. The external parts include a microphone, a speech processor, and a transmitter. The internal parts include a receiver-stimulator and an electrode. Some models include a small headpiece that is worn just behind the ear and contains all the external parts while other models also use body-worn modules that are placed in a shoulder pouch, in a pocket, or worn on a belt. The convenience of the all-in-one headpiece is balanced by shorter life for the batteries used in the smaller units, although systems using rechargeable batteries do solve some of these issues.
Within the headpiece, the microphone picks up sound in the environment. The speech processor converts these sounds into a digital signal. The content of the generated digital signal is determined by the programming of the processor and is complex. It includes information about the pitch, loudness, and timing of sound signals and attempts to filter out extraneous noise. The transmitter converts the digital signals into FM radio signals and sends them through the skin to the internal parts of the implant. The transmitter and the internal parts are kept in correct alignment by using magnets present in both the internal and external parts of the device.
The internal parts are those that are surgically implanted into the patient. The receiver-stimulator is disk-shaped and is about the size of a quarter. It receives the digital signals from the transmitter and converts them into electrical signals. A wire connects the receiver to a group of electrodes that are threaded into the cochlea when the implant is placed. As many as 24 electrodes, depending on the type of the implant, stimulate the ganglion cells in the cochlea. These cells transmit the signals to the brain through the auditory nerve. The brain then interprets the signals as sound.
The sounds heard through an implant are different from the normal hearing sounds and have been described as artificial or robot-like. This is because the implant's handful of electrodes cannot hope to match the complexity of a person's 15,000 hair cells. However, as more electrodes are added, electrode placement issues are solved, and the software for the implant speech processor takes into account more and more aspects of sound, the perceived results are moving closer to how speech and other sounds are naturally perceived.
Despite the benefits that the implant appears to offer, some hearing specialists and members of the deaf community believe that the benefits may not outweigh the risks and limitations of the device. Because the device must be surgically implanted, it carries some surgical risk. Manufacturers can not promise how well a person will hear with an implant. Moreover, after getting an implant, some people say they feel alienated from the
Surgical procedure
The procedure can be preformed on an outpatient basis for adult and adolescent patients. With children, it is often performed with a one-night stay in the hospital.
The internal parts of the implant are placed under the skin behind the patient's ear. The area is shaved, although newer procedures allow for sterilization of the hair in the area so less shaving has to occur. Once the sterile field is established, the surgeon makes an 2–3 in (5–7.6 cm) incision behind the ear and opens the mastoid bone (the ridge on the skull behind the ear) leading into the middle ear. A depression is made in the bone next to the opening to allow the receiver-stimulator to sit flush with the skull surface. After seating, the receiver-stimulator is held in place with a long-lasting suture.
The surgeon then goes through the opening in the mastoid bone to create a new opening in the cochlea for the implant electrodes. The electrode is then very slowly and careful threaded through this new opening. Care is taken during the procedure, and the electrode structure itself is designed to align the electrodes as closely as possible to the ganglion cells, as this allows the electrical signals that function to be less powerful. Once in place, the device is tested to be certain it is working. If all is well, the surgeon then closes up the incision with absorbable sutures, so the area does not need to be revisited to remove the stitches.
The entire operation takes between one and two hours, although the procedure is more complex for younger patients due to the smaller size of their middle ear structures and tends to take longer.
Aftercare
For a short period of time after the surgery, a special bandage is worn on the head during sleep. After about one month, the surgical wounds are healed and the patient returns to the implant clinic to be fitted with the external parts of the device and to have the device turned on and mapped. Mapping involves fine tuning the speech processor and setting levels of stimulation for each electrode, from soft to loud.
The patient is then trained in how to interpret the sounds heard through the device. The length of the training varies from days to years, depending on how well the person can interpret the sounds heard through the device.
Risks
As with all operations, there are risks with this surgery. These include:
- infection at the incision site
- bleeding
- complications related to anesthesia
- transient dizziness
- facial paralysis (rarely)
- temporary taste disturbances
- additional hearing loss
- device failure
However, it should be noted that serious surgical complications have been observed at only one in 10,000 procedures of this type.
Some long-term risks of the implant include the unknown effects of electrical stimulation on the nervous system. It is also possible to damage the implant's internal components by a blow to the head, which will render the device unworkable.
A further consideration is that the use of magnetic resonance imaging (MRI) for patients with cochlear implants is not recommended because of the magnets present in the devices. Several companies have developed implants that do not use magnets or have altered the receiver-stimulator make up to make it easier to remove the magnets before testing. One fact that reduces the concern about MRI testing is that for many medical indications, MRI can be replaced with a computer assisted tomography scan (CAT or CT scan), which is not a problem for persons with cochlear implants.
Additionally, in July 2002, the Food and Drug Administration (FDA) issued a warning about a possible connection between increased incidence of meningitis and the presence of a cochlear implant. This warning included special vaccine recommendations for those with implants, as well as the voluntary removal from the market of certain devices. Specifically, those implants that included a positioner to hold the electrodes in place in the cochlea appear to be associated with an increased risk of the disease.
Normal results
Most profoundly deaf patients who receive an implant are able to discern medium and loud sounds, including speech, at comfortable listening levels. Many use sound clues from the implant, together with speech reading and other facial cues, to achieve understanding. Almost all adults improve their communication skills when combining the implant with speech reading (lip reading), and some can understand spoken words without speech reading. More than half of adults who lost hearing after they learned to speak can understand some speech without speech reading. Especially with the use of accessory devices, the great majority can utilize the telephone with their implants.
Children who were born deaf or who lost their hearing before they could speak have the most difficulty in learning to use the implant. Research suggests, however, that most of these children are able to learn spoken language and understand speech using the implant. In general, the earlier the implant occurs the greater the chance of the implant providing sufficient sound input to provide speech understanding. As with the use of the telephone in adults, accessory devices such as special microphones often help the function of the implant in classroom settings.
Resources
books
Christiansen, John B. and Irene W. Leigh. Cochlear Implants in Children: Ethics and Choices. Washington, DC: Galladet University Press, 2002.
Niparko, John K., ed. Cochlear Implants: Principles and Practices. Philadelphia: Lippincott, Williams & Wilkins, 2000.
periodicals
Cheng, Andre K., et al. "Cost-Utility Analysis of the Cochlear Implant in Children." Journal of the American Medical Association 284, no. 7 (August 16, 2000): 850–856.
"The Earlier the Implant, the Greater the Benefit." The Hearing Journal (February 2001).
Mraz, Stephen J. "Breaking the Wall of Silence." Machine Design (December 9, 1999).
organizations
Alexander Graham Bell Association for the Deaf. 3417 Volta Place NW, Washington, DC 20007. (202) 337-5220. http://www.agbell.org .
Cochlear Implant Club International. 5335 Wisconsin Ave. NW, Suite 440, Washington, D.C. 20015-2052. (202) 895-2781. http://www.cici.org .
Hearing Loss Link. 2600 W. Peterson Ave., Ste. 202, Chicago, IL 60659. (312) 743-1032, (312) 743-1007 (TDD).
National Association for the Deaf. 814 Thayer Ave., Silver Spring, MD 20910. (301) 587-1788, (301) 587-1789 (TDD). http://www.nad.org .
other
Cochlear Implant Recipients may be at Greater Risk for Meningitis. FDA Public Health Web Notification. October 17, 2002 [cited February 23, 2003]. http://www.fda.gov/cdrh/safety/cochlear.html .
Carol A. Turkington Michelle L. Johnson
WHO PERFORMS THE PROCEDURE AND WHERE IS IT PERFORMED?
Cochlear implants are inserted during a surgery performed by a specialist in otolaryngology (ear, nose, and throat medicine). The surgery is performed in a larger hospital, usually as on an outpatient basis, but sometimes with an overnight hospital stay.
QUESTIONS TO ASK THE DOCTOR
- How "normal" will the implant make my/my child's hearing?
- What kind of training does the patient undergo to learn how to use the device?
- How do I care for the device?
jack
the wire from the implant is now coming through his ear drum and has infection coming from his ear. Has anyone had these symptoms and what did they do?
any comments, reassurance or suggestions will be appreciated
thank you
I'm tough (usually) and I was not prepared for this. But one of the reasons of not being prepared was due to the misinformation and data scrubbed responses from people on various forums. I worked in IT and know data scrubbing. When someone commented on a forum (she was afraid of surgery was met with a simple response from me that I was too, was removed by the moderators after sending me a not to remove it... that's all it was. The site was created by one of the three companies here and they obviously didn't want anything remotely negative on it. I found it deceptive and within their right to remove it and I never touched that site again).
It has been a year since receiving the processor gear on the outside of my head. I had to relearn sounds and I'm far from doing that. Since the implant removes sensori-neural AND (I didn't think of this) bone conduction, there's no residual hearing and it's creating issues. The implant isn't flush against my head, so it's too difficult to wear the external piece behind my ear. I wear a clip on my shirt. A side-effect not mentioned is tinnitus. I had it to an extent prior to surgery but it is greatly compounded now. I feel the dreadfully expensive device works better as a mask (to dull some of the tinnitus) rather than an aid to process sounds. I cautioned people about it on a forum about removing comments and again, I was attacked and was told I was lying and I was not. I hold the upper-hand here because again, 30 years in the computer field made it clear to me what was going on.
People say you can wear glasses with it. When I said I could not on one forum, I was attacked. I have wire-frame glasses and had avoided using them. They do not work for me without pain because the implant bulges so much in back of my ear. At this time, I'm waiting to learn if the implant can be surgically altered so I can see (wear glasses). If I had plastic (thicker-frames) glasses, I would be totally lost.
Santhi, Everyone's head and brain is different. I would say it didn't work for me and if I can find out if insurance will pay for the removal, that will happen. No one can say how your dad will be and I'm close to his age. I just caution you to ignore the marketing material and he should understand he will not hear sounds but process them (there's no hearing because sounds are transmitted through the brain) and recognizing music ... I don't know. I can't and music was an integral part of my life. The cost will vary depending on your location, the surgeon, and the hospital. Also, the warranty may only be 3-5 years for something that in my case on one ear for surgery, the device, and audiology adjustments was well over $50k.
There are absolutely people in my age bracket who have had help due to the implants. I'm not one of them (and using the phone is impossible right now).
Please, website masters, allow this to remain. There is so much bad information about implants floating around that it's mind-boggling.
My younger brother is planning to have this surgery very soon, so please urgent response will be highly appreciated.
Thanks.
Do your research before you allow your children to get cochlear implant
The CDC , FDA , New England Journal of Medicine all have reports on the web stating "cochlear implants can cause meningitis in children" had we known this 10 years ago, we would never had opted for the cochlear . Now the dr does not want to remove the implant stating removable will cause spinal fluid leakage . Someone ( Please Lord ) help us !!!
I got a cochlear implant in my 60's & find that I can follow slow speech reasonably well IF I can also speech read - not exactly what I was expecting, but better than relying on the written word for incoming communication!
It accept frequencies that generate pain and other human controls. Or it accept as a receiver of sound and imaging and transmitter to external sources.
Can you help me get rid of it. What are typical expense for such treatment.